Fucoidan, a sulfated polysaccharide extracted from brown algae, has demonstrated potential in preventing some viral and bacterial infections, as well as enhancing antitumor activity in both laboratory and animal studies. Research on fucoidan’s biological activities has been extensive, but its specific roles in immune function and its ability to boost immune responses in living organisms have yet to be comprehensively studied.
So in this blog, I would like to share the following study: “Fucoidan Can Function as an Adjuvant In Vivo to Enhance Dendritic Cell Maturation and Function and Promote Antigen-Specific T Cell Immune Responses” by Jun-O Jin et al. The study investigated the effects of fucoidan on splenic dendritic cell (DC) function and its adjuvant effect in vivo.
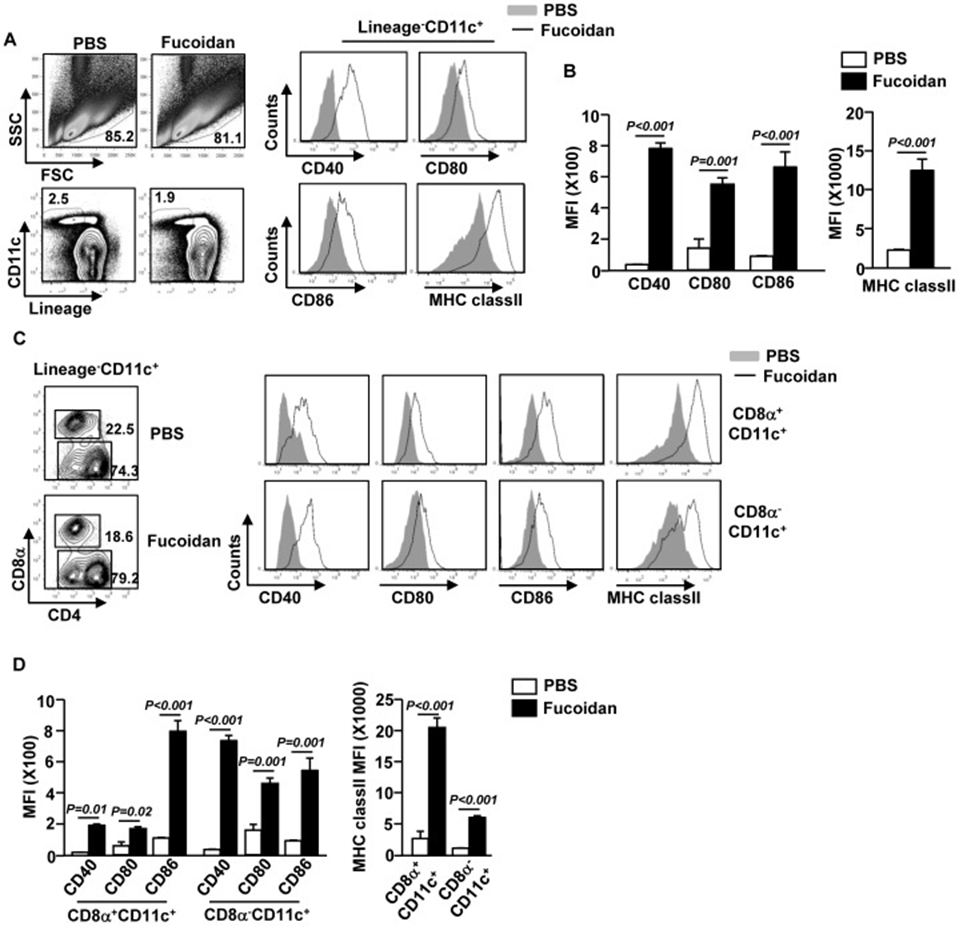
First, they assessed whether fucoidan could also induce the maturation of mouse DCs in vivo. Fucoidan treatment in C57BL/6 mice significantly boosted the expression of CD40, CD80, CD86, and MHC class II on splenic CD11c+ cDCs, as demonstrated in Figure 1A and B. We then investigated the impact of fucoidan on CD8α+ and CD8α− cDC subpopulations 24 hours after fucoidan administration. Fucoidan treatment significantly increased the expression of CD40, CD80, CD86, and MHC class II on both CD8α+ and CD8α− cDCs (Figure 1C and D). This data demonstrates that fucoidan treatment promotes the maturation of splenic cDCs in a live setting.
The study investigated the impact of fucoidan on cytokine production by collecting serum and spleens from C57BL/6 mice three hours post-administration and analyzing them for proinflammatory cytokines. Mice administered fucoidan also showed a dramatic increase in serum IL-6, IL-12p70, and TNF-α levels. Fucoidan treatment significantly increased IL-6, IL-12p70, and TNF-α production in the culture medium. Furthermore, CD11c+ cDCs purified from mice administered fucoidan for 2 h had dramatically higher IL-6, IL-12p40, and TNF-α mRNA levels than control mice. Systemic fucoidan administration resulted in the maturation of splenic cDCs, as shown by the upregulation of costimulatory molecules and the production of pro-inflammatory cytokines.
Since fucoidan-induced maturation of CD8α+ and CD8α− cDCs, they assessed whether fucoidan-induced maturation of splenic cDCs could subsequently promote CD4 and CD8 T cell responses in vivo. According to the study, fucoidan treatment led to a notable increase in splenic CD4 and CD8 T cells producing IFN-γ and TNF-α, the characteristic cytokines of Th1 and Tc1 cells. In comparison, the percentage of splenic IL-17- or IL-4-producing CD4 and CD8 T cells was not increased by fucoidan. Serum levels of IFN-γ and TNF-α were also significantly increased by fucoidan. Furthermore, mice treated with fucoidan had significantly higher amounts of T-bet and IFN-γ, mRNA, key transcription factors for Th1 and Tc1 cells in the spleen than control mice. However, fucoidan treatment did not affect the mRNA levels of GATA3 and RORγt, the key transcription factors for Th2 and Th17 cells.
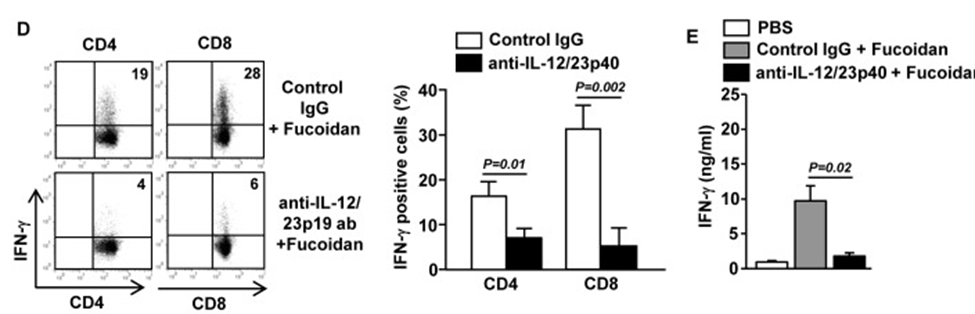
Next, to examine whether the enhancement of Th1 and Tc1 responses by fucoidan was dependent on IL-12, which is the main inducer of Th1 and Tc1 cells in various immune responses, they injected anti-IL-12/23p40 antibody into C57/B6 mice pre-injected with fucoidan or PBS. The promotion effect of fucoidan on IFN-γ production in CD4 and CD8 T cells was almost entirely lost when IL-12/23p40 was neutralized, as demonstrated in Figure 2D. Furthermore, the increase in serum IFN-γ levels by fucoidan was also completely abolished by anti-IL-12/23p40 treatment (Figure 2E). Thus, fucoidan promotes the generation of IFN-γ-producing Th1 and Tc1 cells in an IL-12-dependent manner. These data suggest that fucoidan promotes Th1 and Tc1 responses by enhancing IL-12 production.
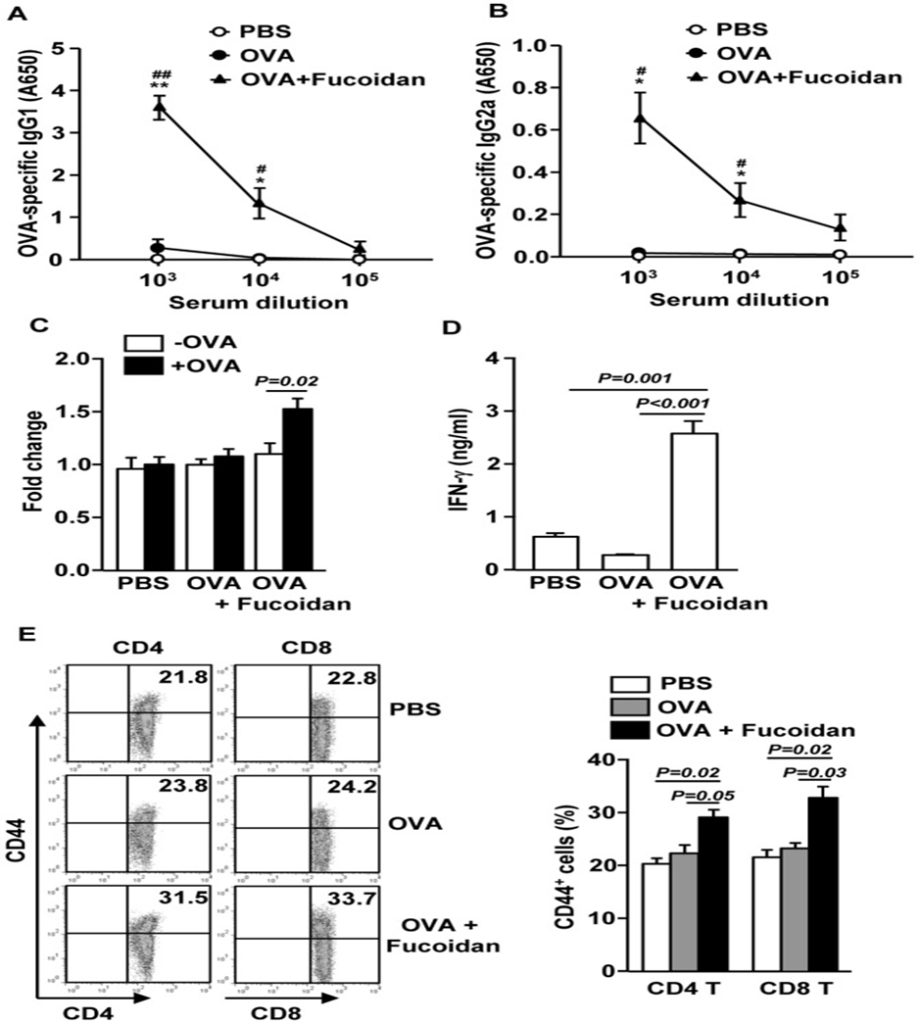
To examine whether fucoidan exerts an adjuvant effect in the body, mice were immunized with OVA and fucoidan, and the specific antibody production and T-cell responses against OVA were examined. C57BL/6 mice were injected intraperitoneally with 10 mg/kg fucoidan. On day 35, serum was analyzed for OVA-specific IgG1 and IgG2a. The data presented in Figure 3A and B show that immunization with OVA plus fucoidan significantly enhanced the production of anti-OVA IgG1 and IgG2a antibodies in mice compared to those immunized with OVA alone. On day 35, splenocytes were also harvested, restimulated with OVA for 4 days in vitro, and analyzed for OVA-induced T-cell responses.
Splenocytes from mice immunized with OVA + fucoidan showed significantly higher cell proliferation and IFN-γ production than splenocytes from control mice immunized with OVA only (Fig. 3C and D). These results indicate that fucoidan may function as an adjuvant by promoting Th-type immune responses. The researchers then investigated the potential of fucoidan to stimulate the development of effector/memory T cells in mice that had been immunized with OVA, by analyzing the surface expression of CD44 on these cells. As shown in Figure 4E, fucoidan injection significantly increased the percentage of CD44+CD4 and CD8 T cells (Figure 3E). These data suggest that fucoidan functions as an adjuvant to enhance antigen-specific T and B cell immune responses.
The study aimed to further demonstrate fucoidan’s adjuvant effect on antigen-specific T-cell responses in vivo by initially determining whether fucoidan could promote antigen presentation or cross-presentation by dendritic cells. Splenic CD11c+ cDCs dramatically upregulated the surface expression of MHC class I and II molecules after treatment with OVA + fucoidan, but not those treated with OVA alone. They then performed adoptive transfer experiments to detect the proliferation of OVA-specific OT-I and OT-II T cells. Mice were injected with PBS, OVA, or OVA + fucoidan 24 hours later. CFSE dilution assay was used to quantify the expansion of OT-I and OT-II cells after three days. The proliferation of OT-I and OT-II T cells was significantly increased in mice immunized with OVA + fucoidan compared with mice immunized with OVA alone. Fucoidan appears to enhance the immune response by acting as an adjuvant that boosts antigen presentation and activates both CD4 and CD8 T cells specific to the presented antigen.
The study concluded that OVA immunization combined with fucoidan as an adjuvant effectively prevented B16-OVA tumor cell growth in mice. The combined results point to fucoidan’s potential as an adjuvant, capable of stimulating Th1 immune responses and CTL activation, thus potentially advancing tumor vaccine development.
Source: PLoS One. 2014; 9(6): e99396. doi: 10.1371/journal.pone.0099396
