Alcoholic liver disease can manifest in individuals who engage in heavy daily alcohol consumption, leading to the accumulation of fat in the liver and subsequent inflammation. Heavy alcohol consumption can result in the development of a life-threatening condition known as severe alcoholic liver disease. If not treated, it can advance to cirrhosis or liver cancer and result in around 3.3 million fatalities annually.
In this blog, I am sharing the following study, “Effect of fucoidan on ethanol-induced liver injury and steatosis in mice and the underlying mechanism” by Meilan Xue et al.
First, all mice (male C57BL/6J mice) were randomly divided into four groups: 1) control group, 2) model group, 3) diammonium glycyrrhizinate-treated group (200 mg/kg body weight) DG group, and 4) fucoidan-treated group (300 mg/kg body weight). Liver injury and steatosis were observed in mice treated with ethanol for 8 weeks.
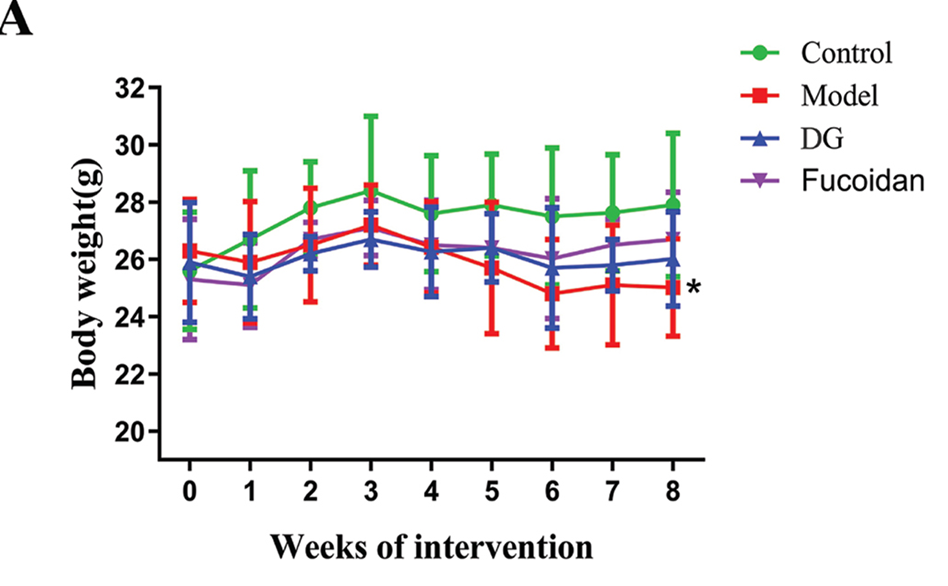
Effects of fucoidan on weight gain, serum aminotransferases, and hepatic inflammatory factors. When comparing the final weights of mice in the DG and fucoidan groups with the model group, it was observed that the weights were slightly higher in the former two groups. However, the difference in weights was not found to be statistically significant, as shown in Figure 1 A. In addition, serum ALT and AST levels, which are major markers of liver damage, were significantly higher in mice with ethanol intake. Mice in the DG group had lower serum ALT and AST levels compared with mice in the model group. In a similar manner, administering fucoidan resulted in a significant 46.8% reduction in serum ALT levels when compared to the mice in the control group. The data indicates that the administration of fucoidan effectively relieved ethanol-induced liver injury, showing a comparable effect to DG.
Also, Ethanol administration increased liver IL-6, IL-1β, and IL-18 levels compared with the control group. Compared with the model group, DG administration reduced liver IL-1β and IL-6 levels, whereas fucoidan administration significantly reduced liver IL-6 and IL-18 levels.
Histopathological observation of the liver did not show any fatty degeneration in the control group. On the other hand, the intake of ethanol caused disorganized hepatic cords, the development of Mallory bodies, the infiltration of inflammatory cells, and a substantial accumulation of lipid droplets in the liver tissue. In comparison to the model group, both the DG and fucoidan groups of mice showed decreased lipid accumulation and reduced inflammatory infiltration.
When examining the liver at a microscopic level, it was observed that hepatocytes in the model group exhibited irregular shapes, an abundance of lipid droplets, malformed mitochondria, and disrupted endoplasmic reticulum. The DG and fucoidan groups showed enhanced hepatocyte morphology, well-organized endoplasmic reticulum, and clear mitochondrial cristae.
The ethanol-fed model group mice showed significantly increased serum TG, CHOL, LDL-CH, and TBA levels compared with the control group mice. However, fucoidan treatment decreased serum CHOL levels and liver TG and TBA levels, and increased liver HDL-CH levels compared to the model group.
Western blot analysis was used to detect the expression levels of oxidative stress markers in liver tissue. Mice in the model group had higher levels of CYP2E1, Grp78, and 3-NT compared with those in the control group. In contrast, the fucoidan group of mice showed notable decreases in CYP2E1, Grp78, and 3-NT levels compared to the mice in the experimental group.
Immunofluorescence analysis revealed that mice that consumed ethanol had higher expression of CYP2E1 compared with those in the control group. There was only a slight presence of CYP2E1 staining in the mouse liver tissues following fucoidan treatment. These results suggest that fucoidan treatment may suppress ethanol-induced liver injury.
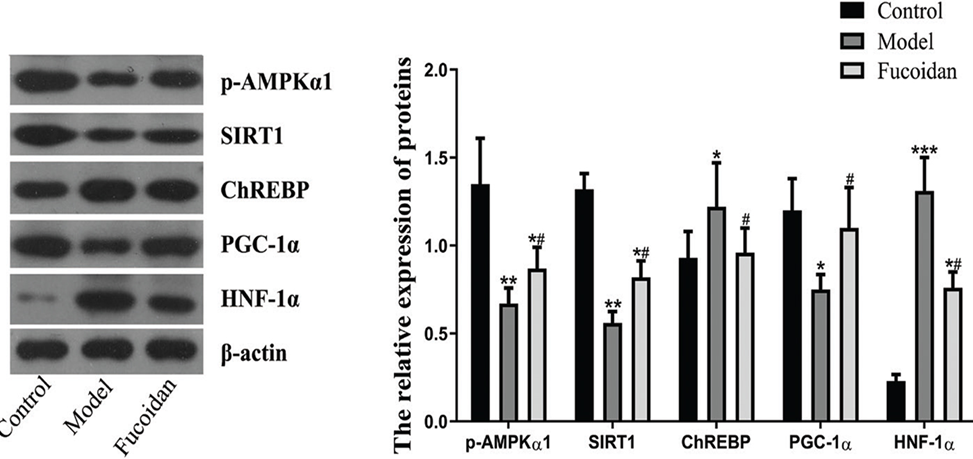
In order to comprehend how fucoidan protects against liver damage and fat accumulation caused by ethanol, we examined the levels of p-AMPKα, SIRT1, ChREBP, PGC-1α, and HNF-1α in the liver. These factors play a crucial role in regulating fat metabolism and inflammation in the liver. Western blot results showed that ethanol feeding suppressed the levels of p-AMPKα, SIRT1, and PGC-1α, thereby promoting alcoholic hepatic steatosis. However, the administration of fucoidan resulted in a reversal of the ethanol-induced changes in the expression levels of p-AMPKα, SIRT1, and PGC-1α, as illustrated in Figure 2. The mice that received ethanol exhibited significantly higher levels of hepatic ChREBP and HNF-1α as opposed to the control mice. On te other hand, the mice treated with fucoidan displayed reduced levels of ChREBP and HNF-1α in comparison to the model mice.
To evaluate the effect of fucoidan on bile acid metabolism, HPLC was used to detect bile acid levels in feces. Mice that consumed alcohol had significantly higher cholic acid (CA) and muricholic acid (MCA) levels and lower deoxycholic acid (DCA), taurodeoxycholic acid (TDCA), and glycodesoxycholic acid (GDCA) levels in feces compared with those in the control group. The administration of Fucoidan resulted in a decrease in CA and MCA levels, while TDCA levels were found to be increased when compared to the model group. These data indicate fucoidan can alleviate ethanol-induced bile acid metabolic disorders.
Fucoidan significantly reversed the reduction in ileal farnesoid X receptor (FXR) and fibroblast growth factor 15 (FGF15) levels induced by alcohol feeding and reduced CYP7A1 (cholesterol 7α-hydroxylase) expression and total bile acid levels in liver tissue. Additionally, fucoidan played a role in regulating the composition of gut microbiota. This led to an increase in Prevotella, while causing a decrease in Paraprevotella and Rhombusia. This was observed through the use of 16S rDNA high-throughput sequencing.
In conclusion, Fucoidan proved its efficacy in mitigating alcohol-induced hepatic steatosis and rectifying bile acid metabolic disorders in mice. Furthermore, it provided substantial protection against alcohol-induced liver damage by activating the AMPKα1/SIRT1 pathway and modulating the gut microbiota-bile acid-liver axis.
Source: Food Nutr Res. 2021; 65: 10.29219/fnr.v65.5384. doi: 10.29219/fnr.v65.5384

I like this internet site because so much useful stuff on here : D.
Thank you, your comment gives me the courage and strength to continue blogging.